PRRC (Person Responsible for Regulatory Compliance)
Under the EU MDR (2017/745) and IVDR (2017/746), medical device manufacturers must designate a Person Responsible for Regulatory Compliance (PRRC) to ensure that all regulatory obligations are fulfilled.
What is a PRRC?
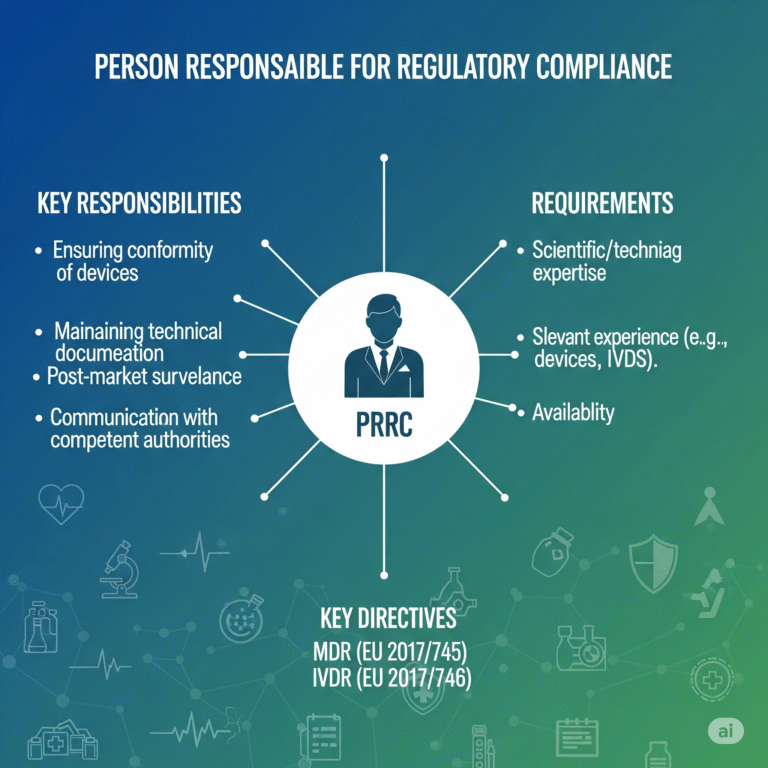
The PRRC (Person Responsible for Regulatory Compliance) is a mandatory role introduced by the EU MDR and IVDR. This individual must possess specific qualifications and is responsible for ensuring that the manufacturer complies with applicable regulations, especially relating to:
- Technical documentation
- Post-market surveillance
- Vigilance reporting
- Regulatory conformity declarations
- Device release procedures
Applicable to manufacturers and authorized representatives operating in the EU, the PRRC plays a vital role in demonstrating commitment to regulatory excellence.

Why is PRRC Service Important?
Appointing a qualified PRRC is not only a legal requirement in the EU — it also:
- Ensures internal accountability and oversight
- Minimizes the risk of non-compliance or regulatory penalties
- Demonstrates a strong quality and regulatory culture
- Supports effective communication with Notified Bodies and Competent Authorities
Our PRRC Services
Lisora offers both in-house and outsourced PRRC solutions tailored to meet your business size and regulatory needs. Our services include:
- Acting as your official PRRC (outsourced model)
- Supporting internal PRRCs with compliance frameworks
- Document review and sign-off assistance
- Monitoring post-market and vigilance systems
- Regulatory training for internal teams
Each device class has specific regulatory requirements based on its risk level.
Why Choose Lisora for PRRC Support?
Qualified Experts
Certified specialists with strong MDR and IVDR experience
Trusted Confidential
Secure handling of all regulatory & data efficiently
Integrated Approach
Smooth coordination with your team and EU reps always
Flexible Options
Full-time or outsourced PRRC services for your company
Frequently Asked Questions (FAQs)
1. Who is required to have a PRRC?
All EU-based medical device manufacturers and authorized representatives under MDR and IVDR must appoint a PRRC.
2. What qualifications must a PRRC have?
A PRRC must have a degree in law, medicine, pharmacy, engineering, or a related field, plus at least one year of experience in regulatory affairs or quality management — or four years of professional experience without a degree.
3. Can we outsource the PRRC role?
Yes. Small and micro-enterprises are permitted to outsource the PRRC role to a qualified third party like LogicSouls.
4. Is one PRRC enough for both MDR and IVDR?
Yes, provided the individual is qualified under both regulations and responsibilities are clearly defined.
5. What happens if we don’t have a PRRC?
Lack of a designated PRRC is a serious non-compliance issue under MDR/IVDR and can lead to penalties or product withdrawal from the EU market.
