MHRA Medical Device Registration (UK)
Ensure your medical or IVD device is fully compliant with the UK’s MHRA regulations. We offer expert guidance on every step of the MHRA registration process from documentation to submission.
What is MHRA Medical Device Registration?
The Medicines and Healthcare products Regulatory Agency (MHRA) is the UK’s official authority responsible for regulating medical devices and ensuring public safety.
After Brexit, the UK introduced its own regulatory pathway that requires medical devices and in-vitro diagnostic devices (IVDs) to be registered with the MHRA before being placed on the UK market.
MHRA registration ensures:
- The device meets UK safety and performance requirements
- The manufacturer is traceable
- Patients and clinicians are protected through proper oversight
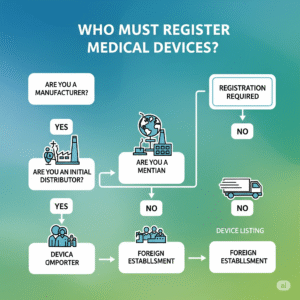
Who Must Register Their Devices?
MHRA registration is mandatory for the following:
- UK-based manufacturers of medical or IVD devices
- Non-UK manufacturers via a UK Responsible Person (UKRP)
- Distributors and importers (if no other party has registered the device)
Without registration, your device cannot be legally marketed in the UK.
What Devices Are Covered?
The MHRA requires registration for a wide range of devices, including:
- General Medical Devices (Class I, IIa, IIb, III)
- In Vitro Diagnostic (IVD) Devices
- Custom-Made Devices
- Active Implantable Medical Devices
- Surgical Instruments, Software-based Devices, etc.
Each device class has specific regulatory requirements based on its risk level.

UKCA, CE & UKNI Markings – What’s the Difference?
| Mark | Region | Purpose |
|---|---|---|
| UKCA | Great Britain | UK-specific conformity marking post-Brexit |
| CE | EU & Northern Ireland | Still required in NI |
| UKNI | Northern Ireland | Used when UK-based notified body is involved |
- UKCA is mandatory for most devices in England, Scotland, and Wales
- CE may still be accepted temporarily or in Northern Ireland
- UKNI applies when UK notified bodies are involved but devices are for NI

Step-by-Step MHRA Registration Process
To register your medical or IVD device in the UK, follow these steps:
- Create an account on the MHRA DORS (Device Online Registration System) Portal
- Appoint a UK Responsible Person (UKRP) if you're a non-UK manufacturer
- Upload your device details, technical documentation, and labeling
- Pay the registration fee (based on device class)
- Submit your application through the portal
- Wait for MHRA approval (typically within 5–10 working days)
What is a UK Responsible Person (UKRP)?
If you're based outside the UK, you must appoint a UK Responsible Person (UKRP) to act on your behalf.
UKRP Responsibilities:
- Register your devices with MHRA
- Hold copies of your technical documentation
- Cooperate with MHRA during audits or investigations
- Report incidents or recalls
Your UKRP must be physically located in the UK.


Required Documentation for Registration
To register a device with MHRA, you'll need the following:
- Declaration of Conformity
- Technical File (including risk analysis, device description, etc.)
- Labeling and Packaging Information
- CE/UKCA Certificates (if applicable)
- GMDN or UDI Code
- Manufacturer's Details and Authorized Representative (if applicable)
All documents must be in English and up-to-date.
Post-Registration Obligations
After registration, the manufacturer or UKRP must:
- Keep technical files updated
- Monitor post-market performance
- Submit reports of adverse events (via MHRA Gateway or ICSR)
- Inform MHRA of any significant changes (e.g. labeling, manufacturer, intended use)
- Renew registrations periodically (as per MHRA guidance)
Failure to comply may lead to suspension or cancellation of registration.

Why Choose Us?
Experienced Team
We've helped 100+ clients register devices with MHRA
Fast Turnaround
Complete all submissions in under Five working days
Complete Support
UKRP setup, document review, labeling compliance
Affordable Packages
Transparent pricing for all startups, agencies and enterprises
Frequently Asked Questions (FAQs)
Is CE marking still accepted in the UK?
Only in Northern Ireland or during limited transition periods in Great Britain. UKCA is now required in GB.
How long does MHRA registration take?
Normally 5–10 working days once the documents and fees are submitted.
Can one UKRP represent multiple devices?
Yes, if authorized by the manufacturer for each.
Do custom-made devices need registration?
Yes, if they are to be placed on the market.
Is MHRA registration needed if the product is already CE-marked?
Yes, CE-mark alone does not suffice in Great Britain post-Brexit.

